FDA & Government News
FDA Approval Advances Basal Insulin
-
By
-
March 27, 2026
-
2 min
By
March 27, 2026
As new contaminants enter agrifood systems, international collaboration and analytical capacity will be key
March 27, 2026

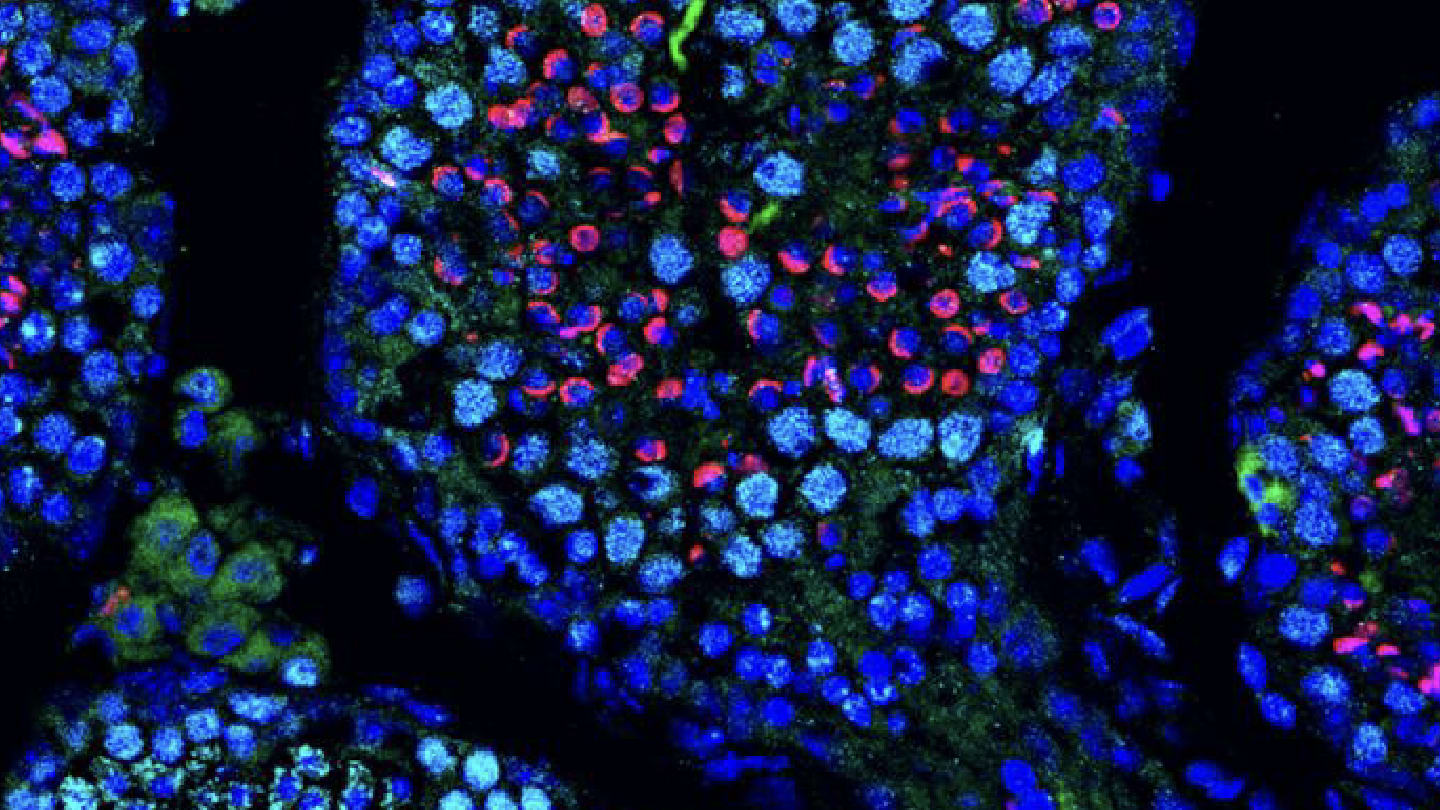
New study indicates that mRNA therapy can restore fertility and sperm production in previously infertile mice
March 27, 2026
A proposed framework highlights potential sources of bias in current estimates
by Kerri Miller
March 26, 2026
