Topcon Introduces Omnia for Automated Eye Pretesting
November 18, 2025
-
2 min
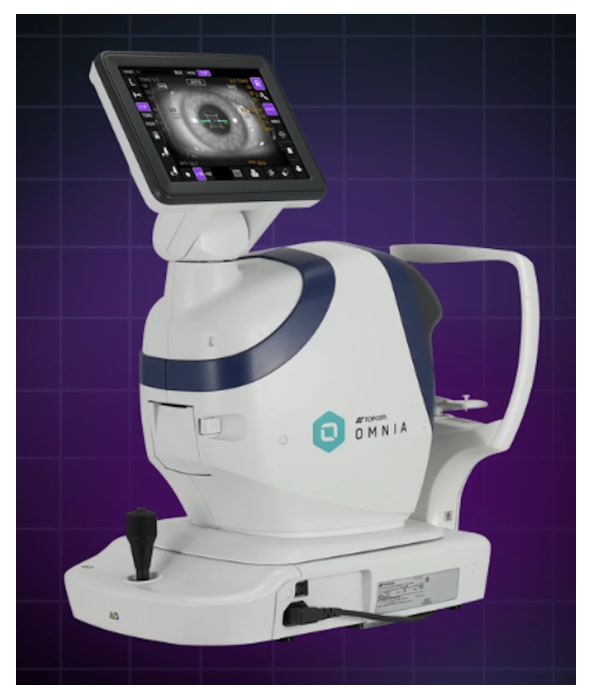
Topcon Healthcare has received FDA 510(k) clearance for Omnia, an innovative 4-in-1 pretesting solution that integrates objective refraction, keratometry, tonometry, and pachymetry. This fully automated device aims to enhance operational efficiency in various clinical environments by consolidating functions, thereby optimizing space and workflow. Features like DICOM compatibility promote seamless data sharing with electronic medical records, while advanced imaging technology and patient comfort enhancements further support reliable output, making it suitable for new users and complex cases alike.
1. Omnia integrates objective refraction, keratometry, tonometry, and pachymetry.2. Designed for efficiency and flexibility in clinical settings.3. Features advanced imaging technology for better measurement accuracy.4. DICOM compatibility enhances data sharing with medical records.5. Automatic alignment function simplifies operation for new users.6. Quieter air puff enhances patient comfort during testing.7. Programmable settings allow customization for practitioners.
Listen Tab content